
Ionization constant (= dissociation constant = self-ionization constant = ion product = autoprotolysis constant ) of water and heavy water, given as function of temperature (☌ and ☏) in figures and tables. Water - Ionization Constant, pKw, of Normal and Heavy Water Online calculator, figures and tables showing heat of vaporization of water, at temperatures from 0 - 370 ☌ (32 - 700 ☏) - SI and Imperial units. Water - Density, Specific Weight and Thermal Expansion Coefficientsĭefinitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360☌ (32 to 680☏). Online calculator, figures and tables giving the boiling temperatures of water in varying vacuum, SI and Imperial units. Water - Boiling Points at Vacuum Pressure Online calculator, figures and tables with melting points of ice to water at pressures ranging from 0 to 29000 psia (0 to 2000 bara). Thermal and thermodynamic properties of ice like density, thermal conductivity and specific heat at temperatures from 0 to -100 oC. Diffusion constants for several gases in water. Gases Solved in Water - Diffusion Coefficientsĭiffusion flux tells how fast a substanse solved in another substance flows due to concentration gradients. Specific volume, enthalpy and entropy of compressed water. Hot and cold water service systems - design properties, capacities, sizing and more.Ĭompressed Water - Properties vs. Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. Temperature - Imperial Units Temperatureĭensities of solids, liquids and gases. Specific gravity (SG) for water is given for four different reference temperatures (39.2, 59, 60 and 68☏).įrom 32 to 212☏ the pressure is 14.7 psi, and for temperatures >212☏, the pressure is equal to water saturation pressure. Specific gravity (SG) for water is given for four different reference temperatures (4, 15, 15.6 and 20☌).įrom 0 to 100☌ the pressure is 1 atm, and for temperatures >100☌, the pressure is equal to water saturation pressure. See also Water Boiling points at high pressure, Boiling points at vacuum pressure, Density, specific weight and thermal expansion coefficient, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Saturation pressure, Specific heat (heat capacity) and Specific volume for online calculatores, and similar figures and tables as shown below. See Water and Heavy Water - thermodynamic properties. Specific gravity of liquid water ranging 32 - 700 o F and 0 - 370 oC is given in figures and tables below: Specific Gravity - SG - is dimensionless and has the same value in the SI system and the Imperial English system (BG). The temperature and pressure must always be specified for both the sample and the reference. Pressure is nearly always 1 atm (101.325 kPa). For gases it is air at room temperature (25☌). Water is normally also used as reference when calculating the specific gravity for solids. related to crude oils and petroleum products. However, also the density at 60 ☏ (15.6☌) or 20 ☌ is often used as the reference temperature, e.g. 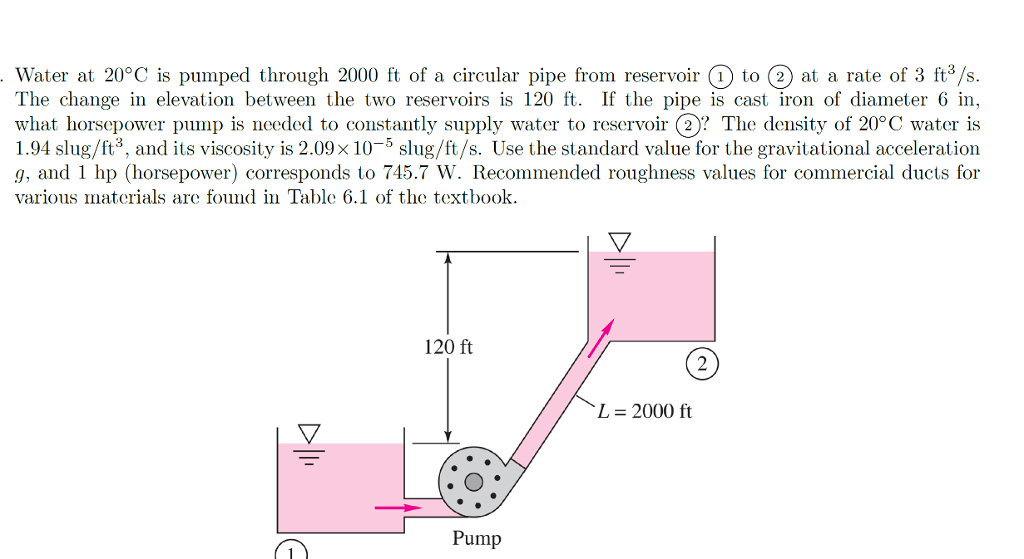
It is most common to use the density of water at 4 oC (39 o F) as a reference since water at this point has its highest density of 1000 kg/m 3 or 1.940 sl/ft 3. Ρ substance = density of the fluid or substance There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials.Specific Gravity (= Relative Density) - SG - is a dimensionless unit defined as the ratio of the density of a substance to the density of a reference substance - at a specified temperature and pressure, and can be expressed as Increases in temperature tend to decrease density since the volume will generally increase. 
An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. 
The calculation of density is quite straightforward.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
